La Bibliothèque d'Applications présente des modèles construits avec COMSOL Multiphysics pour la simulation d'une grande variété d'applications, dans les domaines de l'électromagnétisme, de la mécanique des solides, de la mécanique des fluides et de la chimie. Vous pouvez télécharger ces modèles résolus avec leur documentation détaillée, comprenant les instructions de construction pas-à-pas, et vous en servir comme point de départ de votre travail de simulation. Utilisez l'outil de recherche rapide pour trouver les modèles et applications correspondant à votre domaine d'intérêt. Notez que de nombreux exemples présentés ici sont également accessibles via la Bibliothèques d'Applications intégrée au logiciel COMSOL Multiphysics® et disponible à partir du menu Fichier.
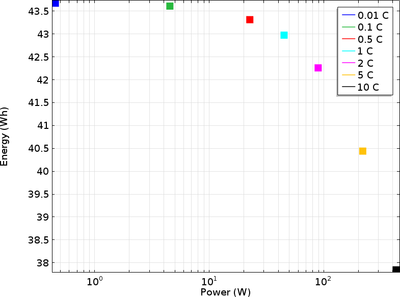
A battery’s possible energy and power outputs are crucial to consider when deciding in which type of device it can be used. A cell with high rate capability is able to generate a considerable amount of power, that is, it suffers from little polarization (voltage loss) even at high ... En savoir plus
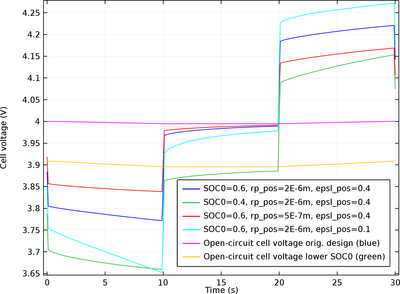
Deposition of metallic lithium on the negative electrode in preference to lithium intercalation is known to be a capacity loss and safety concern for lithium-ion batteries. Harsh charge conditions such as high currents (fast charging) and/or low temperatures can lead to lithium plating. ... En savoir plus
This tutorial digs deeper into the investigation of rate capability in a battery and shows how the Lithium-Ion Battery interface is an excellent modeling tool for doing this. The rate capability is studied in terms of polarization (voltage loss) or the internal resistance causing this ... En savoir plus
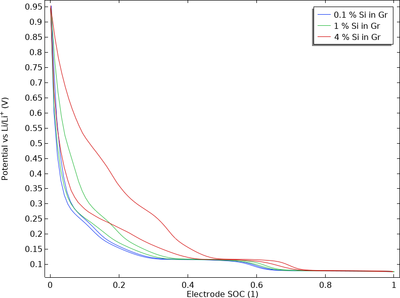
Due to its high capacity, silicon (Si) is often added to graphite in the negative electrode of lithium-ion batteries. Silicon–graphite blended electrodes may exhibit significant thermodynamic voltage hysteresis (“path dependence”) because the equilibrium potential of the lithium–silicon ... En savoir plus
This tutorial demonstrates how to integrate multiple Lumped Battery models into the Electrical Circuit interface. Two batteries are connected in series. Each battery is protected by a shunt resistances that is activated if the battery state-of-charge reaches below a certain threshold ... En savoir plus
This tutorial example models the currents and the concentration of dissolved metal ions in a battery (corrosion cell) made from an orange and two metal nails. This type of battery is commonly used in chemistry lessons. Instead of an orange, lemons or potatoes can also be used. En savoir plus
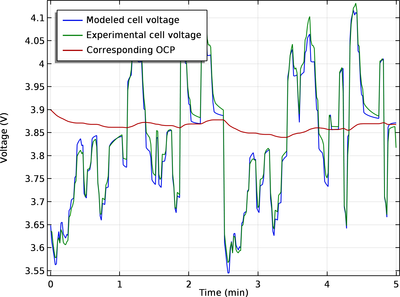
This tutorial uses a “black-box” approach to define a battery model based on a small set of lumped parameters, assuming no knowledge of the internal structure or design of the battery electrodes, or choice of materials. The input to the model is the battery capacity, the initial state ... En savoir plus
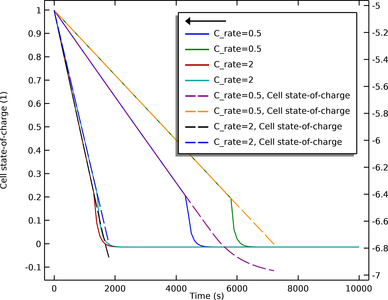
This tutorial demonstrates the Lumped Battery interface for modeling capacity loss in a battery. A set of lumped parameters are used to describe the capacity loss that occurs due to parasitic reactions in the battery, assuming no knowledge of the internal structure or design of the ... En savoir plus
Zinc-Silver oxide (Zn-AgO) batteries are used in different industries due to their high capacity per unit weight. In this work, discharge of a Zn-AgO battery is simulated using the Battery with Binary Electrolyte interface. The electrochemical reactions in the positive and negative ... En savoir plus
This model exemplifies how to compute the internal temperature distribution in a prismatic battery during a high-rate charge. The electrochemistry is described by a a lumped two-electrode model, which is coupled to the heat transfer model. The heat transfer model includes the effects of ... En savoir plus